CYGENTA strictly follow the GMP quality management system
010-89288951
mawenxin@cgtcdmo.com
malan@cgtcdmo.com
Building 1, No. 7 Tiangui Street, Tiangongyuan Street, Daxing District, Beijing


扫码关注
mawenxin@cgtcdmo.com
malan@cgtcdmo.com
Building 1, No. 7 Tiangui Street, Tiangongyuan Street, Daxing District, Beijing

CYGENTA quality control laboratory with a total area of about 1100 square meters, with GMP concept for design, construction, operation and management. It can provide early Pharmaceutical Analysis Services of CGT drugs including plasmid, lentivirus, AAV, CAR-T, CAR-NK, MSCs, iPSC and so on; It can provide services such as development, optimization, qualification/validation, quality research, quality control analysis and stability research of analytical methods in different stages such as IND, clinical trial phaseⅠ, phase Ⅱ, phase Ⅲ and commercialization, etc,To meet the needs of customers in different stages of drug quality.
The Xiji Laboratory includes four functional areas: physical and chemical experimental area, microbiological experimental area, PCR experimental area, and cell experimental area. According to their different functions, each has its own independent air conditioning system and is equipped with the most advanced analytical instruments and equipment at home and abroad, effectively improving detection flow and sensitivity.

Physical and chemical testing area is equipped with regular physical and chemical testing instruments, pharmaceutical water testing equipment, process gas testing equipment, environmental monitoring equipment, raw materials and excipients testing equipment, ultra-high performance liquid chromatograph, gas chromatograph, capillary electrophoresis and other precision physical and chemical instruments.

The microbiology laboratory features a dedicated full-exhaust air handling system and has been officially registered to comply with P2 biosafety laboratory protection standards. Sterility test, Microbial Limit/bioburden test, endotoxin test, mycoplasma test and other biological safety test can be carried out. The sterility test is equipped with two positive pressure double operators isolators. The maximum detection capacity can reach 18 batches at the same time.

The PCR laboratory comprises four segregated sections:the reagent preparation area, the sample processing area, the standard preparation area, and the amplification and analysis area.Through reasonable partition, the possibility of aerosol contamination in PCR experiment is greatly reduced. To avoid cross-contamination, each work zone reagent and sample is delivered through a unidirectional transfer window. Reagent preparation area and sample processing area and CNC area are relatively positive pressure,The other two rooms are relatively negative pressure with the CNC area. This area is equipped with 2 fluorescence quantitative PCR instruments and 1 digital PCR instrument.

The cell culture laboratory is divided into two areas: the cell potency testing area and the positive control testing area. It is equipped with a dedicated full-exhaust HVAC system and contains B2 biosafety cabinets. The laboratory has completed registration to meet P2 biosafety laboratory protective standards. This area can be used for cell killing experiments, cell secretion factor detection, replication lentivirus detection, virus infection titer detection, etc.
CYGENTA strictly follow the GMP quality management system
Standardized management of quality elements such as personnel, premise, facilities and equipment, materials, environmental control, methodology development, qualification and validation could ensure the data reliability
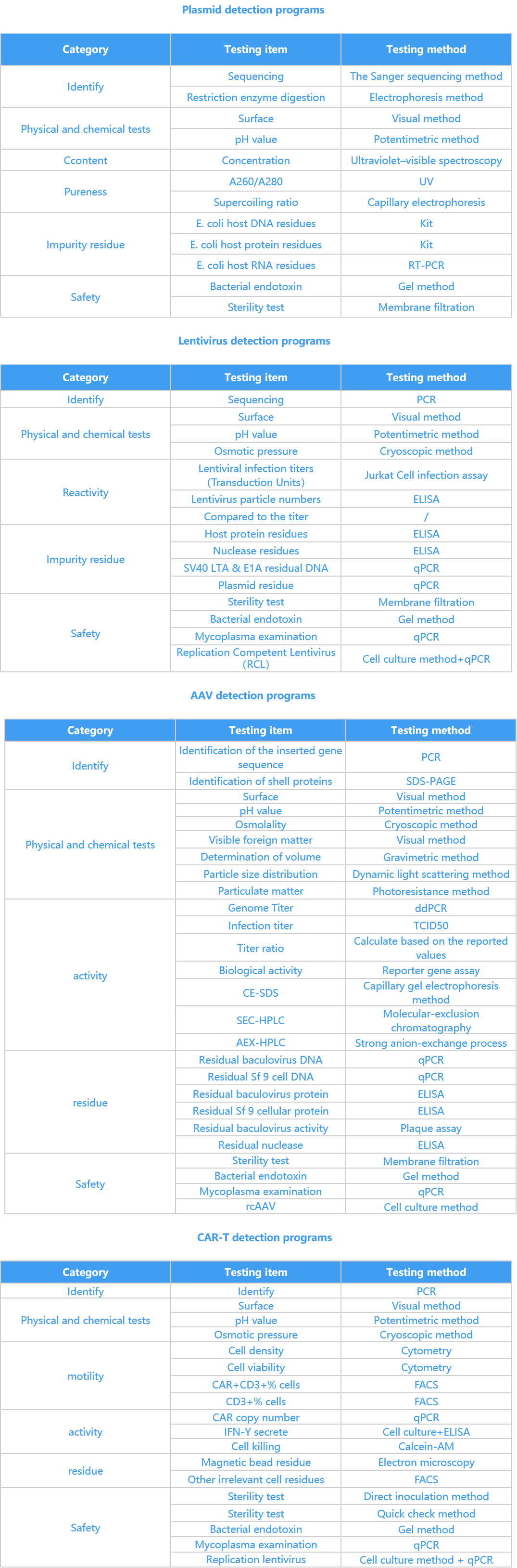
CYGENTA platform has more than 80 analysis methods
We can provide customers with method development and validation services, plasmid, virus, cell and other products release testing, raw material testing and laboratory environmental control services

010-89288951
mawenxin@cgtcdmo.com
malan@cgtcdmo.com
Building 1, No. 7 Tiangui Street, Tiangongyuan Street, Daxing District, Beijing

Official WeChat
official account
Copyright © 2024 SiFotonics, All Rights Reserved. 京ICP备2022016556号-1
